Brief Description
By means of a simple model (consisting of a cup and two thermometers) the structure of the Earth with its atmosphere is simulated. Two thermometers – one being located under the cup that is placed upside down – are illuminated over a certain period of time. This illustrates the effects of an atmosphere on a planet.
This resource is part of the educational kit "The Climate Box". You can read more about the kit in the presentation attached. Find all related resources selecting the category "Our fragile planet" and "secondary level".
Goals
This actvity demonstrates the effects of an atmosphere on the surface temperature of a planet by driving a natural Greenhouse Effect.
Learning Objectives
The pupils carry out a simple analogue experiment that demonstrates the basics of the Greenhouse Effect. They have to set up the experiment according to the instructions and perform temperature measurements. Finally, they have to analyse and interpret the result as well as discuss the relation to the planetary Greenhouse Effect.
Evaluation
The pupils should be able to explain the phenomenon observed in their own wording. They should also be able to record data on their own and draw the necessary conclusion for their environment.
Question 1: The temperature inside a greenhouse is higher than the environmental temperature outside the greenhouse.
Question 2: The earth with its atmosphere is warmer than similar planets with a thinner atmosphere or an atmosphere with less greenhouse gases.
Question 3: The thermometer below the cup eventually shows a higher temperature than the thermometer located outside.
Task:
- The air inside the cup heats up faster than outside. Hence, the thermometer under the cup shows a higher temperature.
- The Earth including its atmosphere is warmer than a similar planet with a thinner atmosphere or on that contains less greenhouse gases.
Question 4:
- the natural Greenhouse Effect: an effect which occurs due to the natural composition of a planetary atmosphere, where no greenhouse gases have been added from outside. This effect is necessary in order make life on Earth possible. A simple calculation shows that without the greenhouse effect the average surface temperature on our earth would be 6°C instead of 15°C.
- the anthropogenic Greenhouse Effect: an artificially generated Greenhouse Effect caused by human intervention which fuels the natural Greenhouse Effect by the additional emission of greenhouse gases like CFC, carbon dioxide, methane, and nitrous oxides.
Materials
The number of items corresponds to the number of students carrying out the experiments. One set consists of:
- Lamp
- Transparent plastic cup
- 2 thermometers
Background Information
Global warming
A key phenomenon of the climate change we witness is the unprecedented speed of rising atmospheric temperatures. Monitoring this quantity is one of the central objectives of the European Copernicus programme which uses ground based in situ measurements as well as satellite based remote sensing techniques.

Figure 1: Averaged global land surface temperature map for March 2016 obtained with the MODIS spectrograph on board NASA’s Terra satellite of the EOS programme. The colour code indicates temperatures between -25°C and +45°C (Credit: NASA Near Earth Observations, http://neo.sci.gsfc.nasa.gov/view.php?datasetId=MOD11C1_M_LSTDA).
An example of what remote sensing can achieve is given in Figure 1, which provides a global map of the land surface temperature averaged over an entire month (March 2016). In order to model the temperature development from the past into the future, the processes of heating the land surface and the atmosphere must be determined.
The Greenhouse Effect
In 1824 the French mathematician and physician Jean-Baptiste Fourier writes in his scientific paper Mémoire sur les températures du globe terrestre et des espaces planétaires about the global thermal budget, an effect which he called effet de serre and which can be translated as Glasshouse or Greenhouse Effect. Since then, Fourier has been regarded as the discoverer of this phenomenon. The Greenhouse Effect can be separated into two components:
- the natural Greenhouse Effect: an effect which occurs due to the natural composition of a planetary atmosphere, where no greenhouse gases have been added from outside. This effect is necessary in order make life on Earth possible. A simple calculation shows that without the greenhouse effect the average surface temperature on our earth would be 6°C instead of 15°C.
- the anthropogenic Greenhouse Effect: an artificially generated Greenhouse Effect caused by human intervention which fuels the natural Greenhouse Effect by the additional emission of greenhouse gases like CFC, carbon dioxide, methane, and nitrous oxides.

Figure 2: Temperature reconstruction for the northern hemisphere during the past 2000 years based on latest measurements (Mann, Zhang, Hughes, et al. 2008, Proceedings of the National Academy of Sciences, Vol. 105, No. 36, pp. 13252-13257, http://www.ncdc.noaa.gov/paleo/pubs/mann2008/mann2008.html).
Figure 2 suggests a strong increase of the anthropogenic Greenhouse Effect between the years 1800 and 2000. With a constant global warming, the consequences for the Earth may be dramatic ranging from melting of the glaciers and the Arctic ice shield which leads to a rise of the sea levels, to severe damages to the ecological system of the Earth.
Due to an increasing concentration of the various greenhouse gases, the proportion of the infrared radiation trapped in the atmosphere is also higher which leads to an aggravation of the greenhouse effect. Measuring the concentration of these greenhouse gases allows us to draw conclusions about the future development of the global warming.
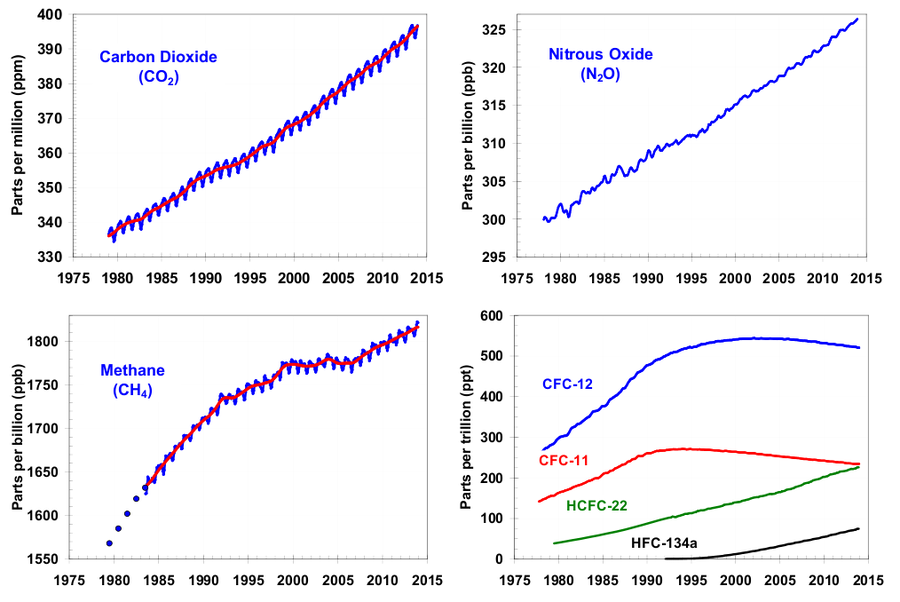
Figure 3: Evolution of the atmospheric abundance of the four greenhouse gases carbon dioxide, methane, nitrous oxide and various CFSs (Credit: NOAA, http://www.esrl.noaa.gov/gmd/aggi/).
The increase of greenhouse gases has been measured with high accuracy (Figure 3). The consequences can be modelled.
Physics of radiation
The air of the atmosphere reflects and absorbs only part of the solar radiation. The rest hits the ground. There it is absorbed and reflected.
Every body with a temperature higher than the absolute zero point emits electromagnetic radiation. The surface of the Earth mainly emits far infrared radiation due to its surface temperature. The frequency distribution of the emitted radiation can be calculated by means of Planck’s Radiation Law. The fraction of radiation of a given frequency and temperature may be written as:

Air - to be more precise the molecules in the mix of gases which we call air – can only absorb light with a certain frequency or wavelength. These are the frequencies which cause the molecules of the air to vibrate. The following diagram shows the vibration modes of carbon dioxide.

Figure 4: Basic vibrations realised as stretching (top) and bending (bottom) of the CO2 molecule (own work).
A molecule is assumed to be IR active, if the vibration modifies the dipole moment. Only molecules that exhibit an electric dipole moment can interact with the incident electromagnetic wave. Hence, the stretching mode to the top left in Figure 4 is IR inactive. Other oscillations modify the dipole moment and are therefore IR active. Generally, an excitation can also occur as a superposition of different vibrational modes.

Figure 5: Absorption spectra of typical greenhouse gases and Rayleigh scattering at atmospheric aerosols (Credit: Robert A. Rohde, The Global Warming Art Project, https://commons.wikimedia.org/wiki/File:Atmospheric_Transmission.png, cropped, https://creativecommons.org/licenses/by-sa/3.0/legalcode).
Molecules with a dipole moment can also be excited to change their rotational state. This leads to the conclusion that air only absorbs a part of the solar spectrum to increase its temperature. The transmissivity of the atmosphere for a given wavelength is given in Figure 6.

Figure 6: Relative atmospheric transmission (Credit: US Government, https://commons.wikimedia.org/wiki/File:Atmosph%C3%A4rische_Absorption.png).
For our experiment this means that air absorbs only a part of the solar radiation hitting the Earth. The remaining radiation that hits the surface is partly absorbed and partly reflected. The following diagram shows the ratio of radiation absorbed by air and land.

Figure 7: Radiation budget of the Earth (Credit: NASA, The Earth Observer. November - December 2006. Volume 18, Issue 6. page 38, after: Kiehl, J. T. and Trenberth, K. E. (1997). "Earth's Annual Global Mean Energy Budget". Bulletin of the American Meteorological Association 78: 197-208, https://commons.wikimedia.org/wiki/File:Keihl_and_Trenberth_%281997%29SunClimateSystem.JPG).
The diagram shows that from the incoming solar radiation of 342 W/m2 only 67 W/m2 (20%) is directly absorbed by the atmosphere. From the remaining portion, 107 W/m2 is reflected back into space. The surface absorbs 168 W/m2, which is a factor of 2.5 more than what the atmosphere absorbs. A large amount of IR radiation is emitted by the ground which interacts with the atmosphere.
Full Activity Description
Question 1: What have been your personal experiences and observations when being in a greenhouse?
Question 2: Certain gases in the atmosphere work in the same way as a glass window of the greenhouse. What would it mean for the Earth, if there would be no atmosphere or an atmosphere with a different composition?
Experimental set-up
- Put one thermometer below the plastic cup that is placed upside down.
- Position the other thermometer next to it.
- Place the lamp in a way so that both thermometers are illuminated.

Figure 8: Experimental set-up (own work).
Question 3: What can you expect after switching on the lamp concerning the temperature with increasing time?
Experimental procedure
Switch on the lamp and observe the temperature inside and outside the cup.
Tasks: Write down your observations during the experiment.
Question 4: In general we differentiate between a natural and an anthropogenic (generated by human beings) greenhouse effect. What is the difference between both effects?
Curriculum
Space Awareness curricula topics (EU and South Africa)
Our fragile planet, atmosphere, climate change
Conclusion
The activity illustrates the basic processes of a Greenhouse Effect by measuring a rise in temperature for two different configurations including a simple analogue of a greenhouse or a planetary atmosphere. In the end, the students will have learned about the difference between a natural and an anthropogenic Greenhouse Effect.

This resource was developed by Marco Türk, Heidelberg University with important contributions by Markus Nielbock, Haus der Astronomie, Heidelberg, Germany. This resource is under peer-review, proof reading, and will be updated and improved in the coming year.

